
The alkali metals have ns 1 valence electron configurations and the lowest electronegativity of any group hence they are often referred to as being electropositive elements. It is so radioactive that studying its chemistry is very difficult. The heaviest element (francium) was not discovered until 1939. However, the group 1 elements, like the group 2 elements, become less reactive with air or water as their atomic number decreases. The potassium burst into flames as soon as it was produced because it reacts readily with oxygen at the higher temperature. Potassium and sodium were first isolated in 1807 by the British chemist Sir Humphry Davy (1778–1829) by passing an electrical current through molten samples of potash (K 2CO 3) and soda ash (Na 2CO 3). Although oxides of both group 1 and group 2 elements were obtained from wood ashes, the alkali metals had lower melting points. Alkali (from the Arabic al-qili, meaning “ashes of the saltwort plant from salt marshes”) was a general term for substances derived from wood ashes, all of which possessed a bitter taste and were able to neutralize acids. The elements of group 1 are called the alkali metals. They have virtually no valence electrons available for bonding or reacting because the full octet of their electron shell is so stable.\) That makes these elements special, known as the noble gases. As we can see, there are four electrons in the 2s and 2p orbitals combined, which together make up the outermost second orbital.Ī noteworthy case is that of group 8: elements in this group have eight electrons, which makes a full octet. To confirm, we check with its electron shell configuration: 1s 22s 22p 2. From this, we infer it has four electrons in the valence shell. 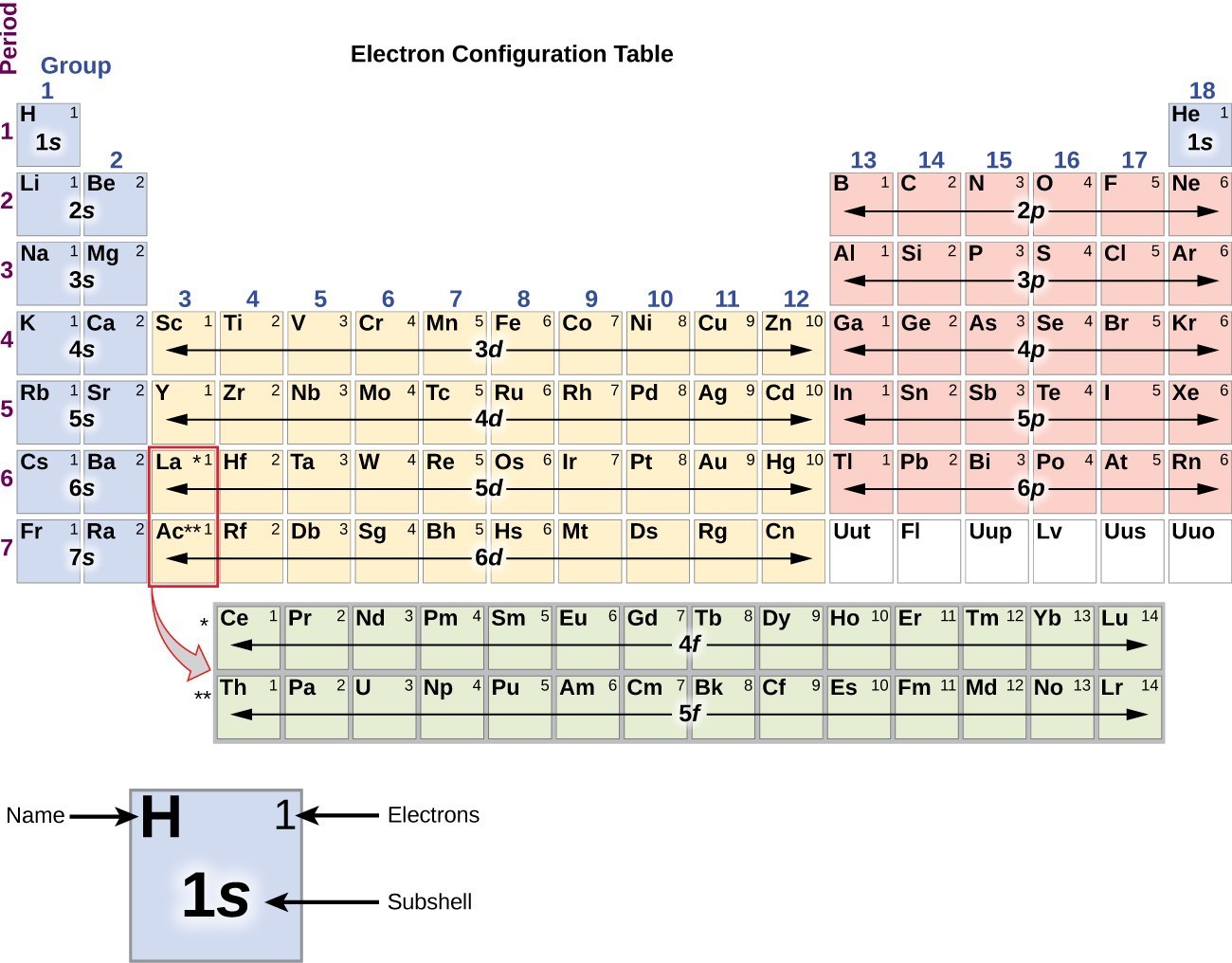
Lastly, we can take a look at carbon, which is in group 4. As we can see, there are seven electrons in the 3s and 3p orbitals combined, which together make up the outermost third orbital. To confirm this, we can take a look at its electron shell configuration: 1s 22s 22p 63s 23p 5. Chlorine is in group 7, indicating it has seven valence electrons. As we can see, there is one electron in the 3s orbital, which is the outermost one.Īnother example is that of chlorine. To confirm this, we can check its electron shell configuration: 1s 22s 22p 63s 1. Examplesįor example, sodium is in group 1, indicating it has one valence electron. In this sense, groups 1 and 2 in the diagram below stay the same, but group 13 is our new “group 3”, group 14 is our new “group 4,” and so on. They are located in the block in the middle of the periodic table. This also means that when looking at a group number, exclude the transition metals. Thus you should take a look at the element’s specific electron shell configuration to figure it out. Transition metals have more complicated electron configurations. Note, however, that this rule only applies to elements that are not transition metals. Look at the group that the element is in, as the group number indicates the number of valence electrons that the element has. You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. How many valence electrons does an element have? On the other hand, nitrogen can form NH 3 so it has a valence of 3, and 3 valence electrons. An element’s valence was historically determined by how many hydrogen atoms it could bond to (which is determined by how many valence electrons it has available for bonding): for example, carbon can form CH 4 so it has a valence of 4, and 4 valence electrons. The term valence refers to the ability of an element to form bonds with other atoms. It is located on the outermost shell (in this case, the shell resembles a ring). This also means that the number of valence electrons that an element has determines its reactivity, electronegativity, and the number of bonds it can form.įor example, in the figure below showing a simplified diagram of sodium’s electron configuration, the valence electron is shown in red. These electrons, being the furthest from the nucleus and thus the least tightly held by the atom, are the electrons that participate in bonds and reactions. Valence electrons are electrons that located in the outermost electron shell of an atom. What are valence electrons? Why are they significant? We will also learn how to tell how many valence electrons an element has. In this tutorial, we learn about valence electrons, what they are, and why they are significant.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
